They are generally very stable chemically and exhibit similar properties of being colorless and odorless. They generally very chemically reactive and are present in the environment as compounds rather than as pure elements. They comprise group 17 of the periodic table, from F through At. The halogen elements are a subset of the nonmetals. The term "nonmetals" is used to classify the elements H, C, N, P, O, S, and Se. Only thorium and uranium are naturally occurring actinides with significant abundance. They, along with the lanthanides, are often called "the f-elements" because they have valence electrons in the f shell. The actinides comprise elements 89 through 103. They, along with the actinides, are often called "the f-elements" because they have valence electrons in the f shell. They are grouped together because they have similar chemical properties. The lanthanides comprise elements 57 (lanthanum, hence the name of the set) through 71. 
They sometimes behave as semiconductors (B, Si, Ge) rather than as conductors. 
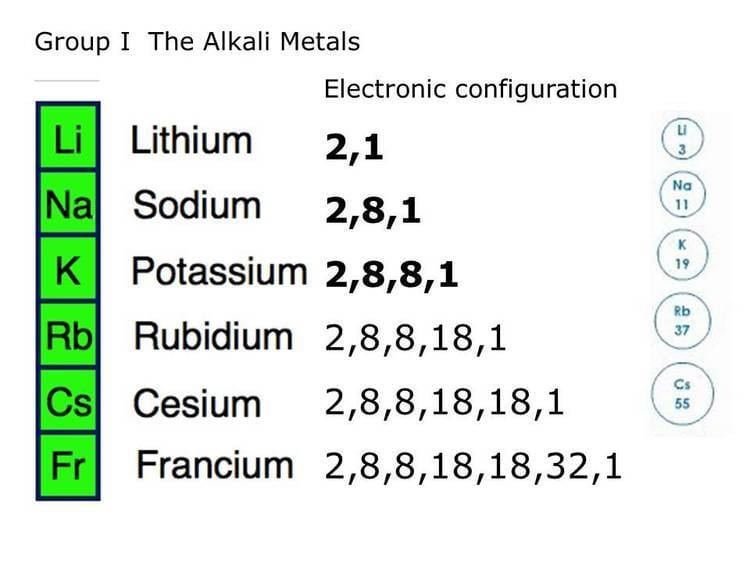
The metalloids are B, Si, Ge, As, Sb, Te, and Po. Metalloid (or "semi-metal" or "poor metal").They tend to be softer and conduct more poorly than the transition metals. The alkaline earth metals or alkaline earths are a set of six elements found in the second group (column) of the periodic table. As their name implies, they have some of the characteristics of the transition elements. The post-transition elements are Al, Ga, In, Tl, Sn, Pb and Bi. The transition elements are metals that have a partially filled d subshell (CRC Handbook of Chemistry and Physics) and comprise groups 3 through 12 and the lanthanides and actinides (see below). Their characteristics are well described and consistent down the group. The alkaline earth metals have very high melting points and oxides that have basic alkaline solutions. The alkaline earth metals make up group 2 of the periodic table, from Be through Ra. Hydrogen is group 1 but exhibits few characteristics of a metal and is often categorized with the nonmetals. They have very similar behavior and characteristics. The alkali metals make up group 1 of the Table, and comprise Li through Fr. The alkali metals are so called because reaction with water forms alkalies (i.e., strong bases capable of neutralizing acids ). In our version of the table, we have chosen the most commonly accepted demarcations between these elements. alkali metal, any of the six chemical elements that make up Group 1 (Ia) of the periodic table namely, lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr). For example, in some tables, Group 12 is is categorized with the post-transition metals, and in others, aluminum and tin are included characterized as Metalloids or poor metals. The dividing line between metals and non-metals is not hard and fast, thus the distinction between "Post-transition metals" and "Metalloids" is represented differently on different versions of the Periodic Table. Metal elements are usually good conductors of both electricity and heat. Wood BJ, Blundy JD (2003) Trace element partitioning under crustal and uppermost mantle conditions: the influences of ionic radius, cation charge, pressure, and temperature.Elements can generally be described as either metals or nonmetals. Lodders K, Fegley JB (1998) The planetary Scientist’s companion. Liang Y, Sun C, Yao L (2013) A REE-in-two-pyroxene thermometer for mafic and ultramafic rocks. Lee C-TA, Luffi P, Chin EJ, Bouchet R, Dasgupta R, Morton DM, Le Roux V, Yin Q-Z, Jin D (2012) Copper systematics in arc magmas and implications for crust-mantle differentiation. Langmuir D (1997) Aqueous environmental geochemistry. Holland HD (1984) The chemical evolution of the atmosphere and oceans. Located on the periodic table in the second column from the right, the halogens are a group of non-metal elements that are not good conductors of heat and electricity. Goldschmidt VM (1937) The principles of the distribution of chemical elements in minerals and rocks. Lithos 77:375–393įrost DJ, Liebske C, Langenhorst F, McCammon CA, Tronnes RG, Rubie DC (2004) Experimental evidence for the existence of iron-rich metal in the Earth’s lower mantle. alkaline earth metal, Any of the six chemical elements in the second leftmost group of the periodic table ( beryllium, magnesium, calcium, strontium, barium, and radium ). Nature 389:842–845Ĭanil D (2004) Mildly incompatible elements in peridotites and the origins of mantle lithosphere. Canil D (1997) Vanadium partitioning and the oxidation state of Archaean komatiite magmas.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
